|
Site-Directed Enzymatic Cleavage Technology: The Precision "Molecular Scalpel" Transforming Genetic Engineering
hits:28 Date:04/12/26
Site-Directed Enzymatic Cleavage Technology: The Precision "Molecular Scalpel" Transforming Genetic Engineering—Backed by ANT BIO PTE. LTD.
1. Concept
Site-directed enzymatic cleavage technology, acclaimed as the "molecular scalpel" of gene editing, enables precise cutting of DNA strands at targeted locations. It stands as a cornerstone in modern molecular biology and genetic engineering, addressing the core challenge of precise DNA manipulation. This technology leverages specialized enzymes to recognize and cleave specific DNA sequences, serving as an indispensable tool in genetic research, biotechnology, medical diagnosis, and therapeutic development.
At its core, the technology relies on two key types of tools: traditional restriction endonucleases and advanced CRISPR-Cas systems. Restriction endonucleases identify specific palindromic DNA sequences (e.g., EcoRI recognizes GAATTC) and cleave phosphodiester bonds at defined positions, generating sticky or blunt ends. The CRISPR-Cas system, by contrast, offers enhanced programmability—guided by single-guide RNA (sgRNA), Cas proteins (such as Cas9) bind precisely to target DNA sequences and induce double-strand breaks (DSBs) via nuclease activity. Unlike restriction endonucleases limited to fixed recognition sites, CRISPR-Cas can target nearly any DNA sequence by designing custom gRNAs.
The standard workflow of site-directed enzymatic cleavage involves three key steps: target sequence recognition (via the enzyme’s binding domain), catalytic cleavage (activated by cofactors like Mg²⁺ to break phosphodiester bonds), and generation of end structures (sticky or blunt ends) that enable subsequent processes such as DNA ligation, cloning, or editing. This precise cleavage capability makes it indispensable for molecular cloning, gene knockout, and sequence modification.
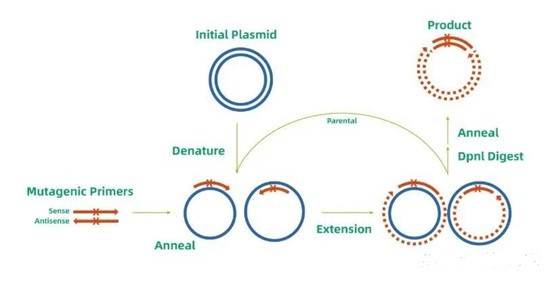
2. Research Frontiers
Site-directed enzymatic cleavage technology is evolving rapidly, with innovations focusing on enhancing precision, expanding functionality, and overcoming practical limitations. One major frontier is the development of next-generation CRISPR tools, such as compact Cas variants (e.g., Cas12f, CasΦ) that offer higher specificity and easier in vivo delivery due to their smaller size. Base editing and Prime Editing systems represent another breakthrough, enabling point mutations (e.g., C→T, A→G) without inducing DSBs, thus minimizing genomic damage and off-target risks.
Integration with artificial intelligence (AI) and multi-omics data is reshaping the field. AI algorithms (e.g., DeepCRISPR) leverage single-cell sequencing and protein engineering data to predict gRNA efficiency and off-target rates, enabling personalized and high-precision experimental design. Automated platforms, such as droplet microfluidics, support high-throughput editing screening, accelerating large-scale functional genomics research.
Clinical translation is a key focus, with CRISPR-based therapies (e.g., CTX001 for β-thalassemia) advancing to late-stage clinical trials. Researchers are also optimizing delivery systems, exploring non-viral methods like lipid nanoparticles (LNPs) and viral vectors (e.g., AAV) to improve targeting and reduce immunogenicity. Additionally, synthetic biology applications are expanding, with tools like Golden Gate cloning using Type IIS restriction endonucleases to enable modular assembly of genetic circuits for industrial biotechnology and metabolic engineering.
Ethical governance and global collaboration are emerging as critical areas, with organizations like UNESCO leading efforts to establish international guidelines for responsible use—particularly for germline editing and gene drives—to address potential ecological and genetic consequences.
3. Research Significance
The significance of site-directed enzymatic cleavage technology spans basic science, translational medicine, and industrial applications. In basic research, it enables precise gene function analysis, allowing researchers to knockout, knock-in, or modify genes to decipher their roles in biological processes and disease pathogenesis. This has accelerated discoveries in fields ranging from developmental biology to cancer research.
In biotechnology, the technology revolutionizes molecular cloning and vector construction, simplifying the creation of recombinant plasmids and genetic constructs essential for protein production, vaccine development, and genetic engineering. In medicine, it has transformed molecular diagnostics (e.g., RFLP technology for detecting genetic mutations in diseases like sickle cell anemia) and paved the way for gene therapies targeting previously untreatable genetic disorders.
For synthetic biology and metabolic engineering, site-directed enzymatic cleavage enables modular assembly of genetic elements, accelerating the design of artificial biological systems for applications such as biofuel production, bioremediation, and industrial enzyme synthesis. By enhancing precision and efficiency, the technology reduces research costs, shortens development cycles, and expands the scope of feasible experiments—ultimately driving innovation across the life sciences.
4. Core Mechanisms, Applications, and Product Support
4.1 Key Mechanisms of Site-Directed Enzymatic Cleavage
4.1.1 Restriction Endonuclease-Mediated Cleavage
Restriction endonucleases recognize specific 4–8 bp palindromic DNA sequences and cleave phosphodiester bonds at precise positions. For example, HindIII recognizes AAGCTT and cuts between the two A bases, producing sticky ends that facilitate DNA ligation. These enzymes are widely used in traditional molecular cloning but are limited to pre-existing recognition sites.
4.1.2 CRISPR-Cas-Mediated Cleavage
The CRISPR-Cas system operates via sgRNA-guided targeting: sgRNA binds to complementary DNA sequences, guiding the Cas nuclease to induce DSBs. Cells repair DSBs through two main pathways: non-homologous end joining (NHEJ), which often introduces small insertions/deletions (indels) leading to gene knockout, and homology-directed repair (HDR), which enables precise insertion of foreign sequences or specific mutations when a donor template is provided.
4.2 Core Application Scenarios
Molecular Cloning and Vector Construction: Site-directed enzymatic cleavage is a foundational step in preparing vectors and insert fragments. Treating vectors (e.g., pUC19) and target genes with the same restriction endonucleases generates compatible ends, which are ligated to form recombinant plasmids. Multiple cloning sites (MCS) in vectors contain multiple enzyme cleavage sites, enabling flexible construct design.
Gene Editing and Functional Research: CRISPR-Cas-mediated cleavage is widely used to study gene function, construct disease models, and conduct genetic breeding. It allows for precise manipulation of genes in cells and organisms, accelerating research on complex diseases and biological pathways.
Medical Diagnosis and Treatment: Restriction fragment length polymorphism (RFLP) technology uses site-directed cleavage to detect gene mutations associated with genetic diseases. CRISPR-based gene therapies are being tested for conditions like β-thalassemia and sickle cell anemia, with promising clinical results.
Synthetic Biology and Metabolic Engineering: Tools like Golden Gate cloning use Type IIS restriction endonucleases (e.g., BsaI) for seamless assembly of multiple DNA fragments, enabling efficient construction of synthetic gene circuits and metabolic pathways for industrial applications.
4.3 Advantages Over Traditional Methods
Precision and Programmability: CRISPR-Cas achieves single-base-level targeting via gRNA design, outperforming traditional restriction endonucleases (which have fixed recognition sites and higher off-target risks). High-fidelity Cas variants (e.g., Cas9-HF1) further reduce off-target effects.
Flexibility and Versatility: CRISPR-Cas only requires gRNA sequence changes to target new sites, eliminating the need to switch enzymes. Fusion of Cas proteins with functional domains (e.g., base editors, activators) expands capabilities beyond cleavage to include gene regulation and epigenetic modification.
Efficiency and Cost-Effectiveness: CRISPR-Cas components can be delivered as plasmids or ribonucleoprotein (RNP) complexes, acting rapidly in cells. Compared to ZFNs or TALENs, CRISPR-Cas has shorter construction cycles and lower costs, making it ideal for large-scale screening.
4.4 Current Challenges and Mitigation Strategies
Off-Target Effects: CRISPR-Cas may cleave partially complementary sequences. Solutions include optimizing gRNA design, using high-fidelity Cas variants, and employing computational prediction tools (e.g., CRISPRoff).
Delivery Barriers: Efficient delivery of enzymes to target cells remains challenging. Viral vectors (e.g., AAV) offer high efficiency but have immunogenicity and capacity limits; non-viral methods (e.g., LNPs) balance toxicity and efficiency.
Variable Repair Efficiency: HDR efficiency varies by cell type and state (e.g., lower in stem cells). Strategies include using repair enhancers (e.g., RS-1) or alternative editing tools like Prime Editing.
Ethical and Regulatory Issues: Germline editing and gene drives raise ethical concerns. International guidelines and interdisciplinary collaboration are addressing these challenges to ensure responsible use.
4.5 How ANT BIO PTE. LTD. Products Support Research
ANT BIO PTE. LTD. provides high-quality reagents tailored to advance site-directed enzymatic cleavage research, leveraging its sub-brands UA (recombinant proteins) and Starter (kits/antibodies) to meet diverse experimental needs.
Key products and their applications include:
UA079022 (S. pyogenes Cas9 NLS): A recombinant Cas9 protein with a nuclear localization signal (NLS), expressed in E. coli. It enables efficient CRISPR-Cas9-mediated gene editing in eukaryotic cells, supporting gene knockout, knock-in, and mutation studies. Its high purity and activity ensure reliable cleavage efficiency, making it ideal for basic research and drug discovery.
S0M1048 (Angiogenesis MiniAb Kit): A specialized kit for studying angiogenesis, a process relevant to cancer and developmental biology. It complements site-directed enzymatic cleavage by enabling functional validation of genes involved in angiogenesis, supporting downstream analysis of edited cell lines or animal models.
S0A0101 (Streptococcus pyogenes SpyCatcher001, His tag): A recombinant SpyCatcher protein expressed in E. coli, used for site-specific protein conjugation. It supports the development of fusion proteins and modified enzymes, enhancing the versatility of site-directed cleavage tools (e.g., engineered Cas proteins for improved targeting).
All ANT BIO PTE. LTD. products undergo rigorous quality control, ensuring high purity, activity, and consistency—critical for generating reliable results in gene editing and molecular biology research.
5. Brand Mission
ANT BIO PTE. LTD. is dedicated to empowering the global life science community with high-quality, innovative biological reagents and solutions. With 15 years of experience in antibody and protein development, the company leverages advanced platforms—including recombinant antibody development (rabbit/mouse monoclonal), recombinant protein expression systems (E.coli, CHO, HEK293, Insect Cells), One-Step ELISA, and PTM Pan-Modification Antibody platforms—to deliver a comprehensive product portfolio.
Through its three specialized sub-brands—Absin (general reagents and kits), Starter (antibodies), and UA (recombinant proteins)—ANT BIO PTE. LTD. adheres to international certifications (EU 98/79/EC, ISO9001, ISO13485) and strict quality standards. The company’s mission is to accelerate scientific discovery by providing tools that enhance experimental precision, efficiency, and reproducibility. ANT BIO PTE. LTD. is committed to supporting researchers in basic science, drug development, and translational medicine, ultimately advancing human health and addressing global challenges in food security and the environment.
6. Related Product List
| Product Code |
Product Name |
| UA079022 |
S. pyogenes Cas9 NLS |
| S0M1048 |
Angiogenesis MiniAb Kit |
| S0A0101 |
Streptococcus pyogenes SpyCatcher001, His tag |
7. AI Disclaimer
This article is AI-compiled and interpreted based on the original work. All intellectual property (e.g., images, data) of the original publication shall belong to the journal and the research team. For any infringement, please contact us promptly and we will take immediate action.
ANT BIO PTE. LTD. – Empowering Scientific Breakthroughs
At ANTBIO, we are committed to advancing life science research through high-quality, reliable reagents and comprehensive solutions. Our specialized sub-brands (Absin, Starter, UA) cover a full spectrum of research needs, from general reagents and kits to antibodies and recombinant proteins. With a focus on innovation, quality, and customer-centricity, we strive to be your trusted partner in unlocking scientific mysteries and driving medical progress. Explore our product portfolio today and elevate your research to new heights.
|