|
Tumor Neoantigen-Based Immunotherapy: Ushering in a New Era of Precision Cancer Treatment
hits:323 Date:03/22/26
1. Literature Information
Research Focus: Exploration of tumor neoantigens as targets for personalized immunotherapy, including neoantigen-specific T-cell therapy and personalized vaccines, along with challenges and future directions in clinical translation.
Core Innovation: Validation of neoantigens (tumor-specific mutated peptides) as ideal targets for precision immunotherapy—demonstrated by clinical successes in T-cell adoptive transfer and personalized vaccines—while highlighting the role of high-quality recombinant proteins in advancing neoantigen research and drug development.
2. Research Background
Tumor neoantigens are specific antigenic peptides derived from somatic mutations in tumor cells. Unlike traditional tumor-associated antigens, they are uniquely expressed in tumor tissues (avoiding autoimmune damage) and recognized by high-affinity T cells (escaping central immune tolerance). Genomic studies confirm that most solid tumors express 1-3+ immunogenic neoantigens, with tumor mutation burden (TMB) positively correlating with neoantigen abundance. This makes neoantigens promising targets for personalized immunotherapy. However, technical bottlenecks—such as inaccurate antigen screening, prolonged production cycles, and inhibitory tumor microenvironments—have hindered widespread clinical application. High-quality recombinant proteins play a critical role in validating neoantigen targets, developing detection assays, and advancing therapeutic strategies.
3. Research Approaches
To advance neoantigen-based immunotherapy, the research team adopted a translational, multi-faceted strategy:
Neoantigen Identification: Using whole-exome sequencing, transcriptomics, and mass spectrometry to screen and validate immunogenic neoantigens from tumor tissues.
Preclinical & Clinical Validation: Evaluating neoantigen-specific T-cell therapy (adoptive transfer) and personalized vaccines (RNA/peptide-based) in clinical trials for various cancers (e.g., cholangiocarcinoma, melanoma).
Efficacy Analysis: Correlating therapeutic outcomes with factors such as T-cell purity, neoantigen specificity, and combination with immune checkpoint inhibitors.
Challenge Resolution: Exploring multi-omics integration (for antigen screening), microfluidic/automated production (for timeliness/cost reduction), and combination therapies (for immune microenvironment modulation).
Tool Development: Utilizing recombinant proteins to support neoantigen detection, vaccine development, and drug screening.
4. Research Outcomes
4.1 Why Tumor Neoantigens Are a Focus of Immunotherapy
Neoantigens offer two core advantages over traditional tumor antigens:
Tumor-Specific Expression: Confined to tumor tissues, eliminating cross-reactivity with normal cells and reducing autoimmune risks.
Immune Recognition: Derived from somatic mutations, they are recognized by high-affinity T cells that escape central tolerance.
Clinical Relevance: TMB correlates with neoantigen number, and most solid tumors express immunogenic neoantigens—providing a foundation for personalized therapy.
4.2 Clinical Potential of Neoantigen-Based Cell Therapy
A landmark 2014 Science study validated the efficacy of neoantigen-specific T-cell therapy:
Patient Case: A 43-year-old advanced cholangiocarcinoma patient (chemotherapy-refractory) had 26 nonsynonymous tumor mutations. CD4+ T cells recognizing an ERBB2IP-derived neoantigen were expanded in vitro.
Therapeutic Outcomes: First infusion (25% specific T cells) achieved tumor shrinkage and 18 months of stability; second infusion (95% specific T cells) resulted in complete remission.
Key Insight: Therapeutic efficacy is closely linked to the purity of infused neoantigen-specific T cells.
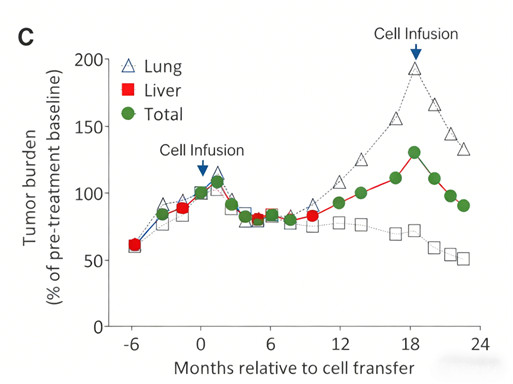
4.3 Precise Immune Activation by Personalized Tumor Vaccines
Two 2017 Nature studies expanded neoantigen applications:
German Team (RNA Vaccine): 13 melanoma patients received personalized RNA vaccines. All developed neoantigen-specific T-cell responses; 8 postoperative patients had prolonged relapse-free survival, 2 advanced patients responded to the vaccine alone, and 1 achieved complete remission with PD-1 inhibitors.
Harvard Team (Peptide Vaccine): 6 patients were treated with peptide vaccines targeting 97 identified neoantigens. 4 remained recurrence-free for 25 months; 2 achieved complete remission (disappearance of lung/subcutaneous metastases) when combined with PD-1 inhibitors upon recurrence.
Synergy: Neoantigen vaccines induce sustained immune memory and enhance the efficacy of immune checkpoint inhibitors.
4.4 Challenges and Solutions in Neoantigen Immunotherapy
| Challenge |
Solution |
| Inaccurate Antigen Screening |
Integrate transcriptome sequencing and mass spectrometry with genomic data to improve immunogenic neoantigen detection. |
| Prolonged Production & High Cost |
Adopt microfluidic chips and automated production to reduce personalized vaccine/cell therapy cycles from 8-12 weeks to 4-6 weeks. |
| Inhibitory Tumor Microenvironment |
Combine with radiotherapy, epigenetic modulators, or chemokine regulators to enhance T-cell infiltration and function. |
4.5 Future Development Directions
Universal Neoantigens: Develop "off-the-shelf" vaccines for high-frequency driver mutations (e.g., KRAS G12D, TP53 R175H) to address personalized therapy limitations.
Combination Therapy Optimization: Explore sequential regimens with CAR-T, bispecific antibodies, or immune checkpoint inhibitors.
Efficacy Prediction Systems: Use T-cell receptor profiling and immune repertoire sequencing to dynamically monitor responses and guide treatment adjustments.
5. Product Empowerment by ANT BIO PTE. LTD.
ANT BIO PTE. LTD.’s UA brand, a specialist in recombinant proteins, provides a key tool for neoantigen and tumor research: the "Human SerpinB3/SCCA Protein" (Catalog No.: UA030001). This high-performance recombinant protein supports multiple aspects of neoantigen-based immunotherapy research.
Key Roles of the Product:
Tumor Marker Research: As a squamous cell carcinoma antigen (SCCA), it enables the development, calibration, and quality control of neoantigen-related immunoassays—supporting accurate neoantigen detection in tumor tissues.
Drug Screening & Evaluation: Serves as a tool molecule for high-throughput screening of neoantigen-targeted antibodies and inhibitors, accelerating therapeutic development.
Research Reliability: High bioactivity (validated by ELISA and cell assays), >95% purity (SDS-PAGE), and endotoxin levels <1.0 EU/μg ensure reliable experimental data.
Versatile Applications: Supports studies in inflammation, immune regulation, and liver diseases—complementing neoantigen research in diverse tumor types.
ANT BIO PTE. LTD.’s portfolio of human recombinant proteins (e.g., APOA1, Myoglobin, Apo-SAA2, ENO-2) further provides comprehensive solutions for multi-parametric tumor research.
6. Brand Mission
ANT BIO PTE. LTD. is dedicated to empowering global life science advancement through three specialized sub-brands: ABSIN (general reagents, ELISA kits), STARTER (antibodies), and UA (recombinant proteins). Leveraging advanced development platforms—including recombinant protein expression (E.coli, CHO, HEK293, Insect Cells), recombinant antibody generation, and One-Step ELISA—we deliver high-quality, compliant products certified by EU 98/79/EC, ISO9001, and ISO13485. Our mission is to partner with innovative pharmaceutical companies, research institutions, and scientists worldwide, providing innovative reagents and solutions that accelerate discoveries in precision oncology, immunotherapy, and personalized medicine.
7. Related Product List
| UA030006 |
Apolipoprotein A-I/APOA1 His Tag Protein, Human |
Host : Human
Expression System : E.coli
Conjugation : Unconjugated |
| UA030005 |
Myoglobin His Tag Protein, Human |
Host : Human
Expression System : E.coli
Conjugation : Unconjugated |
| UA030003 |
Apo-SAA2 His Tag Protein, Human |
Host : Human
Expression System : E.coli
Conjugation : Unconjugated |
| UA030002 |
ENO-2(NSE) Protein, Human |
Host : Human
Expression System : E.coli
Conjugation : Unconjugated |
| UA030001 |
SerpinB3/SCCA Protein, Human |
Host : Human
Expression System : E.coli
Conjugation : Unconjugated |
8. AI Disclaimer
This article is AI-compiled and interpreted based on the original work. All intellectual property (e.g., images, data) of the original publication shall belong to the journal and the research team. For any infringement, please contact us promptly and we will take immediate action.
ANT BIO PTE. LTD. – Empowering Scientific Breakthroughs
At ANTBIO, we are committed to advancing life science research through high-quality, reliable reagents and comprehensive solutions. Our specialized sub-brands (Absin, Starter, UA) cover a full spectrum of research needs, from general reagents and kits to antibodies and recombinant proteins. With a focus on innovation, quality, and customer-centricity, we strive to be your trusted partner in unlocking scientific mysteries and driving medical progress. Explore our product portfolio today and elevate your research to new heights.
|