Home > News > NUZ-001 Preclinical Data Shows Multi-Pathway Protein Clearance Activity
NUZ-001 Preclinical Data Shows Multi-Pathway Protein Clearance Activity
- * New preclinical data show NUZ-001 increases activity of multiple protein clearance pathways in neuronal models
* Demonstrates enhanced activity of both autophagy and proteasomal systems, key processes involved in clearing aggregated proteins
* Supports a differentiated, multi-pathway approach targeting disrupted protein homeostasis in neurodegenerative disease
* Findings further characterise the biological activity of NUZ-001 and strengthen its scientific foundation
* Continues to build evidence supporting NUZ-001 as it progresses through clinical evaluation
MELBOURNE, Australia, April 01, 2026 / Biotech Newswire / -- Neurizon® Therapeutics Limited (ASX: NUZ & NUZOA; OTCQB: NUZTF) ("Neurizon" or "the Company"), a clinical-stage biotech company dedicated to advancing innovative treatments for neurodegenerative diseases, announces new preclinical findings that further characterise the biological mechanism of action for its lead compound, NUZ-001.
The data demonstrate that NUZ-001 and its sulfone metabolite increases activity of multiple protein clearance pathways in neuronal systems. These findings provide additional insight into how NUZ-001 may assist neurons in managing the accumulation of aggregated proteins - a key feature of neurodegenerative diseases such as Amyotrophic Lateral Sclerosis (ALS).
In neurodegenerative diseases, the progressive build-up of abnormal proteins can overwhelm the cell’s natural protein quality-control systems, contributing to neuronal dysfunction and cell death. The new findings show that NUZ-001 increases activity in two critical and complementary pathways involved in maintaining protein balance within neurons: autophagy and the Ubiquitin-proteasome system.
Autophagy supports the bulk degradation of damaged or aggregated proteins, while the proteasome selectively removes smaller protein aggregates. Both pathways play an essential role in maintaining cellular homeostasis. Increasing activity across these systems may help improve the ability of neurons to process and clear aggregated proteins.
This multi-pathway activity represents a differentiated approach compared to therapies that focus on a single pathway and may support a broader restoration of cellular protein balance.
Study
These findings were supported by experiments conducted in established neuronal models designed to assess protein clearance pathways in physiologically relevant human cellular models. Preclinical studies measured key markers of autophagy, including the proteins p62 and LC3 - commonly used indicators of how effectively cells are clearing aggregated proteins - and observed reductions in both following NUZ-001 treatment (Figure 1). These changes are consistent with increased autophagic flux and more efficient degradation of protein aggregates.
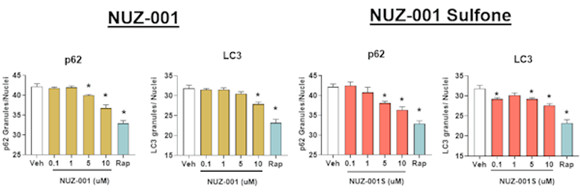
Figure 1: NUZ-001 and NUZ-001 Sulfone decrease p62 and LC3 protein levels in human iPSC-derived neurons. Immunoreactivity of p62 and LC3 was quantified and normalized to total number of nuclei. * p<0.05 vs vehicle (Veh) control using a one-way ANOVA with Dunnett’s post hoc analysis. Rapamycin (Rap) was used as a positive control for autophagy activation
In parallel, NUZ-001 induced an increase in proteasomal activity (Figure 2), providing direct functional evidence of activation of a second protein clearance pathway. Together, these results demonstrate that NUZ-001 activates multiple, endogenous neuronal pathways critical for clearance of misfolded or aggregated proteins.
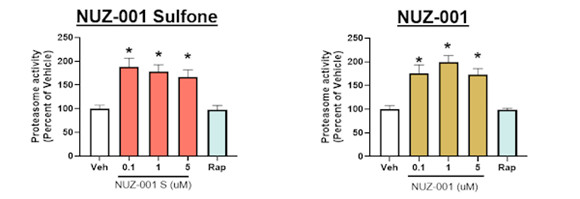
Figure 2: NUZ-001 and NUZ-001 Sulfone increase proteasomal activity in human iPSC-derived neurons. Proteasomal activity was measured using the Proteasome GloTM assay and activity normalized to vehicle control. *p<0.05 vs vehicle (Veh) control using a one-way ANOVA with Dunnett’s post hoc analysis. Rapamycin (Rap) was used as a comparator.
These findings further expand the understanding of the biological activity of NUZ-001 and its potential relevance in neurodegenerative disease. Impairment of protein clearance pathways is a recognised feature of diseases such as ALS, contributing to the accumulation of protein aggregates leading to neuronal damage.
By increasing activity across both autophagy and proteasomal systems, NUZ-001 demonstrates a broader effect on pathways involved in protein homeostasis. This approach may support neuronal resilience and offers a differentiated profile compared to single-pathway strategies.
Interim Executive Chair, Mr Sergio Duchini said: “These findings provide additional insight into the biological activity of NUZ-001 and reinforce our understanding of how it may support key cellular processes disrupted in neurodegenerative diseases.
The observed activity across multiple protein clearance pathways is particularly important, as impairment in these systems is widely recognised as a central feature of diseases such as ALS.
While these results are preclinical, they contribute to the growing body of evidence supporting NUZ-001 as we continue its evaluation in the HEALEY ALS Platform Trial.”
These results strengthen the scientific foundation of NUZ-001 and support its continued development as a potential therapy targeting underlying disease biology across ALS and related neurodegenerative disorders.
Semantic keywords: Neurodegenerative Diseases; Amyotrophic Lateral Sclerosis; Motor Neuron Disease; Proteasome Endopeptidase Complex; Protein Aggregates; Proteostasis; Ubiquitins; Sulfones; NUZ-001; preclinical data; protein clearance; autophagy; proteasomal systems; aggregated proteins; protein homeostasis; Neurizon Therapeutics Limited; sulfone metabolite; ALS; protein accumulation; neuronal dysfunction; cell death; Ubiquitin-proteasome system; cellular homeostasis; protein balance; autophagic flux; protein degradation; proteasomal activity; misfolded proteins; iPSC-derived neurons; proteasome
About Neurizon Therapeutics Limited
Neurizon Therapeutics Limited (ASX: NUZ) is a clinical-stage biotechnology company dedicated to advancing treatments for neurodegenerative diseases. Neurizon is developing its lead drug candidate, NUZ-001, for the treatment of ALS, which is the most common form of motor neurone disease. Neurizon’s strategy is to accelerate access to effective ALS treatments for patients while exploring the potential of NUZ-001 for broader neurodegenerative applications. Through international collaborations and rigorous clinical programs, Neurizon is dedicated to creating new horizons for patients and families impacted by complex neural disorders. NUZ-001 is an investigational product and is not approved for commercial use in any jurisdiction.
Neurizon® is a registered trademark of Neurizon Therapeutics Limited
This announcement has been authorised for release by the Board of Neurizon Therapeutics Limited.
Contacts
Neurizon Therapeutics
Lidija Damjanovic
Marketing & Corporate Affairs
lidija@neurizon.com
+61 (0) 425 700 504
Australia Investor Relations
Henry Jordan
Six Degrees Investor Relations
henry.jordan@sdir.com.au
+61 (0) 431 271 538
U.S. Investor Relations
Matthew Selinger
Integrous Communications
mselinger@integcom.us
+1 415-572-8152
Related News
- How Does Rab10 (Thr73) Regulate Membrane Trafficking and Signaling of the M4 Mus 5/29/2026
- Tecan Establishes Direct Sales and Specialist Lab Automation Service Operations 5/28/2026
- Extensive Range of Independently Verified Cell Lines 5/28/2026
- Flow Chemistry Without the Guesswork: Free New Calculators from Asynt Flow 5/28/2026
- Graphene Quantum Dots Show Promise in Targeting Parkinson's-related Protein clum 5/26/2026
- Targeting NF-κB p65 DNA-Binding Activity: Can Sesquiterpene Lactones’ α-M-γ-B Co 5/26/2026
- NF-κB Signaling Pathway: Generation and Regulatory Mechanisms of p105/p50 5/25/2026
- Everest Medicines Receives Shareholding Increase from Substantial Shareholder CB 5/25/2026
- LECT2: A Liver-Secreted Factor Regulating Renal Fibrosis via Endoplasmic Reticul 5/24/2026
- Immune Checkpoint Antibodies: The Cornerstone Raw Materials of Tumor Immunothera 5/23/2026


