Home > News > Monoclonal Antibodies: Achieving Precise Targeted Therapy Through Structural Optimization and Functional Design
Monoclonal Antibodies: Achieving Precise Targeted Therapy Through Structural Optimization and Functional Design
- 1. Concept
Monoclonal antibodies are specific immunoglobulins produced by a single B lymphocyte clone, characterized by high homogeneity and epitope specificity. Their basic "Y"-shaped structure consists of two heavy chains (~50 kDa) and two light chains (~25 kDa) linked by disulfide bonds, with a total molecular weight of approximately 150 kDa. Functionally, they are divided into two key regions: the Fab segment (responsible for antigen recognition via complementarity-determining regions/CDRs) and the Fc segment (mediating immune effector functions such as antibody-dependent cellular cytotoxicity/ADCC, antibody-dependent cellular phagocytosis/ADCP, and complement-dependent cytotoxicity/CDC). This structural partitioning provides a foundation for antibody engineering, enabling precise optimization for targeted therapy.
2. Research Frontiers
2.1 Basic Structure and Functional Characteristics of Monoclonal Antibodies
The structure-function relationship of monoclonal antibodies defines their therapeutic potential:
* Fab Segment: CDR amino acid sequences determine antigen-binding specificity, enabling precise recognition of target molecules (e.g., tumor antigens, cytokines).
* Fc Segment: Interacts with Fc receptors on immune cells (NK cells, macrophages) and the complement system to activate effector functions, amplifying antitumor or anti-inflammatory effects.
* Structural Flexibility: The hinge region between Fab and Fc segments enhances antigen-binding accessibility, while disulfide bonds ensure structural stability.
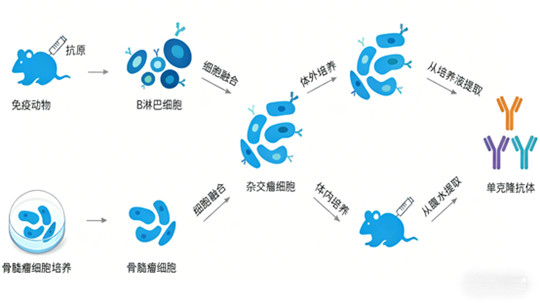
2.2 Evolution of Monoclonal Antibody Preparation Technology
Monoclonal antibody technology has advanced through four key stages, reducing immunogenicity and improving biocompatibility:
* Murine Monoclonal Antibodies: Produced by mouse B cell-myeloma cell fusion, but limited by severe human anti-mouse antibody (HAMA) reactions.
* Chimeric Monoclonal Antibodies: Combine murine Fab segments with human Fc segments (~67% humanization), significantly reducing immunogenicity.
* Humanized Monoclonal Antibodies: Retain only murine CDR regions via CDR grafting (~90% humanization), minimizing immune responses.
* Fully Human Monoclonal Antibodies: Obtained through phage display or transgenic mouse technology, achieving complete human sequences and optimal biocompatibility.
2.3 Mechanisms of Action for Targeted Therapy
Therapeutic monoclonal antibodies exert effects through direct and indirect mechanisms:
* Direct Effects: Act as agonists or antagonists to modulate target activity, inducing apoptosis or blocking oncogenic signaling pathways (e.g., HER2 inhibition in breast cancer).
* Indirect Effects:
ADCC: Fc segment binds to NK cell FcγRIIIa, triggering target cell lysis.
ADCP: Fc segment interacts with macrophage FcγRIIa, promoting tumor cell phagocytosis.
CDC: Activates the complement system to form membrane attack complexes, lysing target cells.
Synergistic Effects: Combined direct and indirect mechanisms provide multi-level intervention for complex diseases.
2.4 Antibody Engineering for Therapeutic Optimization
Antibody engineering tailors molecules to specific therapeutic needs:
* Naked Antibodies: Retain natural structures, suitable for signaling pathway modulation (e.g., immune checkpoint inhibitors).
* Conjugated Antibodies: Link to cytotoxic drugs (antibody-drug conjugates/ADCs) or radionuclides (radioimmunoconjugates/RICs) for targeted drug delivery.
* Bispecific Antibodies: Recognize two distinct epitopes (e.g., tumor antigen and T cell receptor), bridging immune cells to tumor cells for enhanced cytotoxicity.
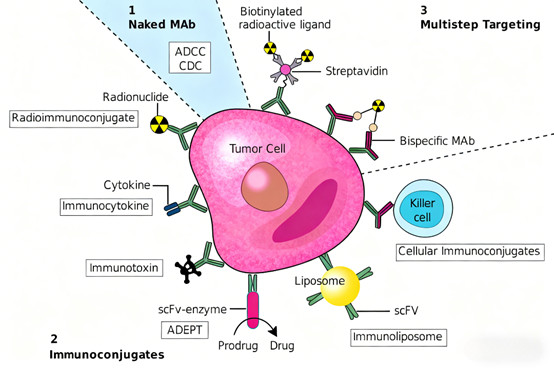
2.5 Clinical Resistance Mechanisms and Implications for Antibody Design
Resistance to antibodies like rituximab provides insights for next-generation optimization:
* Pharmacokinetic Factors: Individual differences in drug metabolism or anti-drug antibody formation reduce exposure.
* Target Alterations: CD20 downregulation or structural variations impair antibody binding.
* Effector Mechanism Impairment: Complement inhibitor upregulation or Fc receptor polymorphisms weaken ADCC/ADCP.
* Tumor Microenvironment Changes: CXCR-4/Gal-1 dysregulation alters immune cell infiltration.
* Apoptosis Pathway Dysregulation: BCL-2 family overexpression or NF-κB activation resists cell death.
* Optimization Strategies: Fc segment engineering (enhancing Fc receptor binding) and dual-target design to overcome resistance.
2.6 Future Directions in Antibody Drug Development
Antibody drug development is evolving toward greater precision and versatility:
* Multifunctional Integration: "Smart antibodies" combining blocking, activating, and immune-recruiting functions.
* Precision Modulation: Epitope-selective design to fine-tune signaling pathway activity.
* Combination Therapies: Synergistic use with chemotherapy, immunotherapy, or targeted drugs to improve efficacy.
* AI-Assisted Engineering: Artificial intelligence accelerates antibody design, optimizing affinity, stability, and specificity.
3. Research Significance
Monoclonal antibodies represent a cornerstone of precision medicine, addressing unmet needs in cancer, autoimmune diseases, and infectious diseases. Their high specificity reduces off-target toxicity, improving therapeutic index compared to traditional treatments. Technological advancements in humanization and engineering have expanded their applicability, while insights into resistance mechanisms drive continuous optimization. As multifunctional therapeutic platforms, monoclonal antibodies bridge basic research and clinical translation, offering new hope for patients with complex diseases. Their development reflects the biopharmaceutical industry’s shift toward targeted, personalized therapy.
4. Related Mechanisms, Research Methods, and Product Applications
4.1 Mechanisms
Antibody-mediated targeted therapy relies on two core mechanisms:
* Antigen-Specific Binding: CDR regions enable precise recognition of disease-related targets (tumor antigens, cytokines, receptors).
* Effector Function Activation: Fc segment interactions with immune cells or complement amplify therapeutic effects, eliminating abnormal cells or modulating immune responses.
4.2 Research Methods
Key methods for antibody development and evaluation include:
* Antibody Generation: Hybridoma technology, phage display, and transgenic mouse systems for antibody discovery.
* Functional Assays: In vitro tests (binding affinity, neutralization activity) and in vivo models (xenografts, autoimmune disease models) to validate efficacy.
* Pharmacokinetic/Pharmacodynamic (PK/PD) Analysis: Evaluating drug absorption, distribution, metabolism, and efficacy in preclinical and clinical studies.
* Resistance Mechanism Studies: Genomic and proteomic analysis to identify resistance drivers and guide antibody optimization.
4.3 Product Applications
ANT BIO PTE. LTD.’s monoclonal antibodies—exemplified by the "In Vivo Anti-Mouse IFNγ Recombinant Monoclonal Antibody" (Catalog No.: S0B0956)—are high-performance tools for preclinical research:
* Core Advantages: High in vivo neutralizing activity, ultra-low endotoxin (<1.0 EU/mg), and superior biocompatibility.
* Key Application Scenarios:
Th1/Th2 Immune Balance Research: Neutralizing IFN-γ to study its role in infections, tumors, and autoimmunity.
Autoimmune Disease Models: Therapeutic studies in experimental autoimmune encephalomyelitis (EAE) and collagen-induced arthritis (CIA).
Infection Immunity: Investigating IFN-γ’s role in intracellular pathogen clearance (e.g., Listeria, Toxoplasma).
Tumor Immune Microenvironment Modulation: Exploring IFN-γ’s impact on tumor immune editing and immunotherapy efficacy.
5. Brand Mission
ANT BIO PTE. LTD. is dedicated to empowering the global life science community with high-quality, innovative biological reagents and solutions. Leveraging advanced development platforms—including recombinant rabbit monoclonal antibody, recombinant mouse monoclonal antibody, rapid monoclonal antibody, and multi-system recombinant protein expression platforms (E.coli, CHO, HEK293, Insect Cells)—and adhering to rigorous international certifications (EU 98/79/EC, ISO9001, ISO13485), we strive to deliver reliable, performance-proven tools that accelerate scientific breakthroughs in immunology, oncology, and translational medicine. Our commitment to quality and innovation aims to support researchers and clinicians in advancing human health through precise targeted therapy.
6. Related Product List
| Catalog No. | Product Name | Host |
| S0B0956 | Invivo anti-mouse IFNγ Recombinant mAb | Rat |
| S0B0998 | Invivo anti-mouse NK1.1 Recombinant mAb | Mouse |
| S0B1070 | Invivo anti-Mouse IL-6 Recombinant mAb | Rat |
| S0B1101 | Invivo anti-mouse CTLA-4 (CD152) mAb | Armenian hamster |
7. AI Disclaimer
This article is AI-compiled and interpreted based on the original work. All intellectual property (e.g., images, data) of the original publication shall belong to the journal and the research team. For any infringement, please contact us promptly and we will take immediate action.
ANT BIO PTE. LTD. – Empowering Scientific Breakthroughs
At ANTBIO, we are committed to advancing life science research through high-quality, reliable reagents and comprehensive solutions. Our specialized sub-brands (Absin, Starter, UA) cover a full spectrum of research needs, from general reagents and kits to antibodies and recombinant proteins. With a focus on innovation, quality, and customer-centricity, we strive to be your trusted partner in unlocking scientific mysteries and driving medical progress. Explore our product portfolio today and elevate your research to new heights.
Related News
- Longbio Pharma Opens Public Offering: Premium Pipeline in Autoimmune & Allergic 6/2/2026
- Collaboration to Improve Sustainable Laboratory Workflows 6/2/2026
- What Are the Regulatory Mechanisms and Functions of RIPK1 Kinase Activity (Ser16 6/1/2026
- A New Opportunity for Regenerative Medicine in Asia 6/1/2026
- Regulatory Mechanisms and Biological Functions of the PI3K Signaling Pathway 5/31/2026
- How Does Clausenamide Alleviate Parkinson's Disease Neuroinjury via Regulating t 5/30/2026
- Key Risks in Medical Device Development 5/30/2026
- How Does Rab10 (Thr73) Regulate Membrane Trafficking and Signaling of the M4 Mus 5/29/2026
- Tecan Establishes Direct Sales and Specialist Lab Automation Service Operations 5/28/2026
- Extensive Range of Independently Verified Cell Lines 5/28/2026


