Reagents:
Molecular Biology
Biochemistry
Cell Biology
ELISA / Diagnostic Kits
Antibody
Serum/Medium
Other Reagents
- Mouse IL-6 ELISA Kit

- Product Detail
- Company Profile
Product Specification
| Usage | Need to bring your own test equipment 1. Microplate reader (can measure the absorption value of 450nm detection wavelength and 540nm or 570nm correction wavelength) 2. High precision liquid dispenser and disposable suction head 3. Distilled water or deionized water 4. Washing bottle (spray bottle), multi-channel plate washer or automatic plate washer 5. 500mL cylinder One, preparation before the experiment 1. Sample collection and storage ① Cell culture supernatant: particles should be removed by centrifugation; Test the samples immediately. If the sample is not tested in time after collection, it is recommended that the sample be divided according to the dosage and stored in the refrigerator at -20 ° C to avoid repeated freezing and thawing. Samples may need to be diluted (1×) Dilute. ② Serum: Samples were collected using a serum separation tube (SST) and samples were left at room temperature for 30 minutes. The samples were centrifuged at 1000g for 15 min. Serum was immediately removed and tested immediately. If the sample is not tested in time after collection, it is recommended to repack according to a single dosage and freeze in ≤ -20℃ refrigerator to avoid repeated freezing and thawing. Samples may need to be diluted (1×) Dilute. ③ Plasma: Plasma was collected using EDTA, heparin, or citric acid as an anticoagulant, centrifuged at 1000g for 15 min within 30 min of collection, and tested immediately. If the samples are not detected in time after collection, it is recommended to separate the samples according to the single dosage and freeze them in &le. -20℃ refrigerator to avoid repeated freezing and thawing. Samples may need to be diluted (1×) Dilute. 2 Reagent preparation (Place all reagents and samples at room temperature for 15 minutes before use. It is recommended that all experimental samples and standards do double hole detection ) 1× Preparation of washing solution: concentrated washing solution in the kit is 20× Mother liquor, diluted to 1× with distilled water before use; Working liquid. Example: Take 10mL concentrated washing solution +190mL distilled water to 200mL, the actual operation can be calculated first, then make up. ②1× Dilution with buffer preparation: concentrate dilution in the kit with buffer 10× Mother liquor, diluted to 1× with distilled water before use; Working solution. example: Take 3mL of concentrated dilution with buffer +27mL of distilled water to a constant volume of 30mL. In practical operation, the required dilution buffer can be calculated according to the dilution multiple of the sample, and then the preparation can be made. ③ Detection of antibody: the dry powder was centrifuged to the bottom of the tube, and 110uL dilution buffer (1×) was used. Dissolve and let stand at room temperature for 5 minutes to obtain 100× Mother liquor; Dilute to 1× before use; Working solution. Calculate the desired volume by using 100uL per well. example: 10 Wells were used, then take 10uL of 100 times the working concentration of the test antibody, using dilution buffer (1×) Constant volume to 1mL, get 1mL of 1× The working concentration of the detected antibody. ④SA-HRP: SA-HRP is 40× Mother liquor, use dilution buffer before use (1×) Dilute to make 1× Working solution, 100uL required per well. example: used 10 holes, then take 25uL of 40× Mother liquor +975uL dilution buffer (1×) Constant volume to 1mL to obtain 1× of 1mL; The working concentration of the detected antibody. ⑤ Chromogenic agent: according to 100uL per well, calculate the amount needed for this test, take out the corresponding volume of chromogenic agent, avoid light; The removed chromogenic agent is only used on the same day. ⑥ Standards: lyophilized standards with dilution buffer (1×) Redissolve, redissolve volume 1000uL, to obtain a concentration of 1000pg/mL standard mother liquor. Gently shake for at least 5 minutes, it is fully dissolved. Add 300uL dilution buffer (1×) to each dilution tube. . The standard mother liquor is diluted according to the picture below, and each tube must be fully mixed before pipetting to the next tube. The standard mother liquor without dilution can be used as the highest point of the standard curve (1000pg/mL), and the dilution buffer (1×) Can be used as the zero point of the standard curve (0pg/mL). 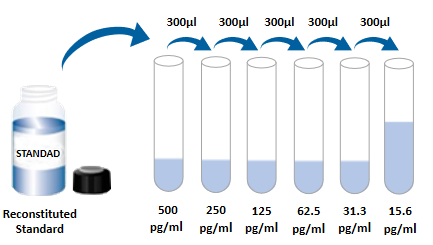 2, operation steps 1. Prepare all required reagents and standards; 2 Remove the microplate from the sealed bag that has been balanced to room temperature, and put the unused slat back into the aluminum foil bag and re-seal it; 3. Add 300uL washing solution to the microplate, let it soak for 30 seconds, discard the washing solution and pat the microplate dry on absorbent paper, please use immediately do not let the microplate dry; 4. Add different concentrations of standard, experimental samples or quality control into the corresponding Wells, 100uL for each well. The Wells were sealed with plate adhesive and incubated at room temperature for 2 hours. 5 Suck the liquid out of the plate and wash the plate using a bottle washer, a multi-channel plate washer, or an automatic plate washer. Add 300uL of washing liquid to each well, and then suck the washing liquid out of the plate. Repeat 3 times. Every time you wash the plate, try to absorb the residual liquid to help you get a good test result. At the end of the last plate wash, please blot all the liquid in the plate or invert the plate and pat all the residual liquid in the absorbent paper; 6. Add 100uL detection antibody to each microwell. Seal the reaction Wells with sealer tape and incubate for 2 hours at room temperature; 7. Repeat the plate washing operation of step 5; 8. Add 100 ULSA-HRP to each microwell and incubate for 20 minutes at room temperature. Be careful to avoid light; 9. Repeat step 5 to wash the plate; 10. Add 100uL of color development solution to each microwell, incubate at room temperature for 5-30 minutes, pay attention to avoid light; 11. Add 50uL of termination solution to each microwell, and the color of the solution in the well will change from blue to yellow. If the color of the solution turns green or the color change is inconsistent, tap the microplate to mix the solution evenly; 12. Within 30 minutes after the termination solution is added, the absorbance value at 450nm is measured using a microplate reader and 540nm or 570nm is set as the correction wavelength. If the dual wavelength correction is not used, the accuracy of the results may be affected; 13 Calculation results: The corrected absorbance values (OD450-OD540/OD570), the compound reading were averaged for each standard and sample, and then the average zero standard OD value was subtracted. Standard curves were created by 4-parameter logic (4-PL) curve fitting using computer software. Alternatively, a curve can be generated by plotting the logarithm of the concentration of the standard against the logarithm of the corresponding OD value, and the best fit line can be determined by regression analysis. This process produces an adequate but less accurate fit to the data. If the sample is diluted, the concentration should be multiplied by the dilution.  3. Kit parameters 1. Recovery rate: Different levels of Mouse IL-6 were incorporated into the cell culture medium samples and the recovery rate was determined. Recoveries ranged from 70 to 114%, with an average recovery of 91%. 2. Sensitivity: The minimum detectable dose (MDD) of Mouse IL-6 was generally less than 1.8pg/mL. The lowest detectable value of mouse IL-6 was calculated as the corresponding concentration based on the average of the zero absorbance values of 20 standard curves plus two standard deviations. 3. Calibration: The ELISA kit was calibrated with high purity recombinant Mouse IL-6 protein expressed in E. coli. 4. Linearity: Four different samples were mixed with high concentrations of Mouse IL-6, followed by dilution (1×). Linearity was determined by dilutes the samples to the detection range.
5. Specificity: This ELISA can detect native and recombinant Mouse IL-6 protein. The following factors in diluent mixture 50 ng/mL (1 x) to detect the concentration and Mouse IL - 6 cross reaction. Interference with Mouse IL-6 was tested by incorporating 50ng/mL of the interfering factor into the intermediate range of recombinant Mouse IL-6 controls. No significant cross-reactivity or interference was observed.
| |||||||||||||||||||||||||||
| Theory | Double antibody sandwich enzyme-linked immunosorbent assay was used in this kit. Specific anti-mouse IL-6 antibody was precoated on a high-affinity microplate. The standard substance, test sample and biotinylated detection antibody were added into the microplate Wells. After incubation, the IL-6 present in the sample combined with the solid-phase antibody and detection antibody to form immune complexes. After washing to remove not combined with material, by adding horseradish peroxidase labeled chain mildew avidin (Streptavidin - HRP). After washing, adding chromogenic substrate, dark color. The reaction was terminated by adding the termination solution, and the absorbance value was measured at 450nm wavelength (reference correction wavelength 540nm or 570nm). | |||||||||||||||||||||||||||
| Composition |
| |||||||||||||||||||||||||||
| Background | Interleukin-6 (IL-6) is a multifunctional cytokine with α-helical structure, 22-28kD phosphorylation and different degrees of glycosylation. It plays an important role in the acute phase of disease response, inflammation, hematopoiesis, bone metabolism and cancer progression. Mature mouse IL-6 has 187 amino acids and is 42% and 85% identical to human and rat IL-6, respectively. Alternative splicing within IL-6 results in a variety of isoforms, some of which exhibit antagonistic properties. Expression of IL - 6 cells is known, "These include CD8+T cells, fibroblasts, synoviocytes, adipocytes, osteoblasts, megakaryocytes, endothelial cells (under the influence of endothelin), sympathetic neurons, cerebral cortical neurons, adrenal medullary chromaffin cells, retinal pigment cells, mast cells, keratinocytes, Langerhans cells, fetal and adult astrocytes, and neutrophils. , monocytes, eosinophils, colon epithelial cells, B1B cells and islet beta cells. The production of IL - 6 is usually in the sugar cortical hormone, catecholamine and under the control of secondary sexual steroids, generally associated with cell activation. The level of IL-6 in normal human blood is in the range of 1pg/mL. It is slightly increased in the menstrual period, moderately increased in the middle and late stage of some cancers, and significantly increased after major surgery. IL-6 elicits cell signaling through a cell surface receptor, which is a heterodimeric complex consisting of a ligand-binding subunit (the IL-6 receptor) and a signal transduction subunit gp130. IL-6 binds to the IL-6 receptor, triggering the binding of IL-6 receptor to gp130 and the dimerization of gp130. gp130 is also a component of the receptors for CLC, CNTF, CT-1, IL-11, IL-27, LIF, and OSM. Soluble IL - 6 receptor is produced by alternative splicing and protease cutting. "Through a trans-signaling mechanism, the combination of soluble IL-6 and IL-6 receptor triggers a response in cells that lack the IL-6 receptor on the surface but express gp130." Expression of the IL-6 receptor is largely restricted to hepatocytes, monocytes, lymphocytes, and resting lymphocytes, and since the gp130 molecule is expressed very broadly, trans-signaling enables a broader range of cell types to respond to IL-6. "Soluble gp130 spliceosome prevented IL-6/IL-6R trans-signaling, but not signaling by other cytokines using gp130 as a co-receptor." The acute inflammatory response induced by IL-6, together with tumor necrosis factor-α (TNF-α) and IL-1, plays an almost unique role in fever and acute liver inflammation. It also plays an important role in the transformation from acute inflammation to acquired immunity or chronic inflammatory diseases. IL-6 dysregulation can promote chronic inflammation, such as obesity, insulin resistance, inflammatory bowel disease, inflammatory arthritis, and sepsis, often involving IL-6 trans-signaling. In the presence of transforming growth factor TGF-β, IL-6 plays an important role in the differentiation of Th17 inflammatory cells from naive T cells. IL-6 regulates bone resorption by promoting the activity of Th17 inflammatory cells, which is the main factor causing inflammatory joint damage in rheumatoid arthritis. IL-6 is involved in the formation and destabilization of atherosclerotic plaques. However, IL-6 also has anti-inflammatory effects, such as secretion of IL-6 by skeletal muscle during physical exercise, promoting hematopoiesis as a growth factor for hematopoietic stem cells, inducing the maturation of B cells into plasma cells, and immortalization of multiple myeloma cells. Also promote IL - 6, but may not start the other inflammation associated with cancer, such as colitis associated cancer. | |||||||||||||||||||||||||||
| General Notes | 1. Please use the kit within the validity period. 2. Components of different kits and different batch kits should not be mixed. 3. If the sample value is greater than the highest value of the standard curve, the sample should be diluted with (1×) The samples were diluted and retested. "If the cell culture supernatant sample needs to be diluted in a distributed manner, cell culture medium may be used for intermediate dilutions, except for the last step when diluent is used." 4. The difference of the test results can be caused by a variety of factors, including the operation of the experimenter, the use of the pipettor, the washing technique, the reaction time or temperature, the storage of the kit, etc. 5. The termination solution in the kit is acidic. Please protect your glasses, hands, face and clothes when using it. 6. For scientific research only, not for in vitro diagnosis. | |||||||||||||||||||||||||||
| Storage Temp. | Unopened kit, 2-8° C Storage | |||||||||||||||||||||||||||
| Test Range | 15.6pg/mL-1000pg/mL |
bio-equip.cn
-
AntBio is a biotechnology group company dedicated to serving life sciences, aiming to help scientists accelerate research and improve work efficiency. AntBio provides comprehensive and high-quality reagent tools for basic research, drug development, and diagnosis, including research grade antibodies, proteins, biochemical reagents, and assay kits. These research tools are widely used in different segments of life science research. The group company currently consists of three brands, Absin, Starter-Bio and UA-Bio.
| Request Information |
| Other Products |
| Related Products |
| Recently viewed products |
- SiteMap



