|
Erythropoietin: Unveiling Physiological Mechanisms, Clinical Potential, and Research Support
hits:26 Date:04/09/26
1. Concept
Erythropoietin (EPO), alternatively called erythrocyte-stimulating factor, is a glycoprotein with a molecular weight spanning 25,000 to 45,000 Da. As a pivotal regulator of erythropoiesis, it occupies a central position in the human hematopoietic system. First identified as an endogenous compound back in 1906, the natural form of EPO has a molecular weight of roughly 34,000 Da and consists of 165 amino acids. Under normal physiological conditions, EPO is predominantly synthesized by interstitial cells in the renal cortex, with a small fraction produced by the liver.
Recombinant human EPO (rhEPO), crafted through genetic recombination technology, has a molecular weight of 30,400 Da. Its physicochemical properties and biological functions closely mirror those of endogenous EPO, despite its gene being located on chromosome 7. Thanks to advancements in bioengineering, EPO preparations have been incorporated into China's National Basic Medical Insurance Directory, emerging as a crucial therapeutic agent for anemia.
2. Research Frontiers
Current research on EPO is advancing on multiple fronts, expanding beyond its traditional role in hematopoiesis. One key area of focus is exploring the non-hematopoietic effects of EPO, including its protective roles in various tissues and organs. Basic research has revealed EPO's cardioprotective properties, such as inhibiting cardiomyocyte apoptosis, mitigating myocardial damage induced by oxidative stress and ischemia, and counteracting reperfusion injury. Additionally, emerging studies highlight EPO's potential in promoting neuronal proliferation and differentiation, regulating the female reproductive system, and safeguarding the retina and bones.
In the clinical realm, research is ongoing to optimize EPO's application in treating cancer-related anemia, with a focus on balancing efficacy and safety—particularly addressing concerns about its potential to stimulate Vascular Endothelial Growth Factor (VEGF) secretion in tumor cells. Furthermore, advancements in recombinant technology are aimed at enhancing the biological activity, stability, and pharmacokinetic profiles of EPO preparations, paving the way for more targeted and efficient therapies. Another active research direction is investigating the dynamic changes of endogenous EPO levels in special populations, such as kidney transplant recipients, to refine personalized treatment strategies.
3. Research Significance
EPO's significance in both basic research and clinical practice is profound. In the context of hematology, it serves as a cornerstone for understanding the regulatory mechanisms of erythropoiesis, providing insights into the body's response to hypoxia and anemia. Clinically, EPO has revolutionized the treatment of renal anemia, a common complication of chronic renal failure, significantly improving patients' quality of life by restoring red blood cell production and oxygen-carrying capacity.
Beyond renal anemia, EPO plays a vital role in managing cancer treatment-related anemia, alleviating symptoms such as fatigue and improving exercise tolerance in chemotherapy patients. Its emerging non-hematopoietic functions open new avenues for treating cardiovascular diseases, neurological disorders, and other conditions, offering potential therapeutic solutions for unmet medical needs.
From a research perspective, high-quality EPO reagents are indispensable for advancing studies on hematopoietic mechanisms, drug development, and disease model construction. Reliable recombinant EPO products enable researchers to conduct precise experiments, accelerating the discovery of novel therapeutic targets and the development of innovative drugs. Moreover, understanding EPO's pharmacokinetic characteristics and safety profiles ensures the rational use of EPO in clinical settings, minimizing risks and maximizing therapeutic benefits.
4. Relevant Mechanisms, Research Methods, and Product Applications
4.1 Physiological and Pharmacological Mechanisms of EPO
4.1.1 Physiological Mechanism of Action
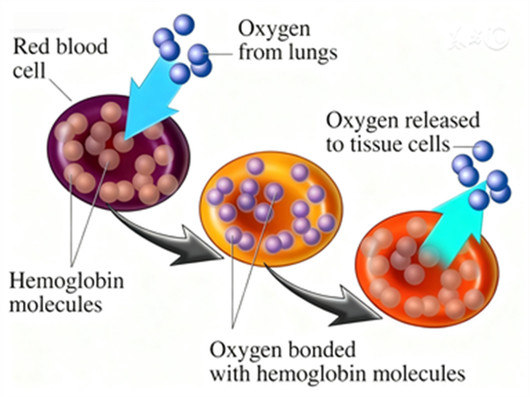
Under physiological hypoxia, the kidneys produce erythropogenin, an enzyme that converts liver-synthesized erythropoietinogen into biologically active EPO. EPO binds to specific receptors on the surface of bone marrow Colony-Forming Unit-Erythroid (CFU-E) or earlier Burst-Forming Unit-Erythroid (BFU-E) cells, triggering a cascade of cellular responses: promoting the differentiation of primitive blood cells into proerythroblasts, accelerating the mitosis of nucleated red blood cells, enhancing hemoglobin synthesis, and facilitating the release of reticulocytes and mature red blood cells from the bone marrow into the bloodstream. As red blood cell counts increase and oxygen-carrying capacity improves, elevated tissue oxygen partial pressure suppresses the activity of relevant enzymes in the liver and kidneys via negative feedback, precisely regulating erythropoiesis and maintaining hematopoietic homeostasis.
4.1.2 Pharmacological Properties
Recombinant human EPO shares identical biological activity with endogenous EPO, directly acting on the bone marrow hematopoietic microenvironment to promote and regulate the proliferation and differentiation of erythroid progenitor cells. In healthy individuals, plasma EPO concentrations are maintained at 10–30 U/L, but can surge a hundredfold to a thousandfold during tissue hypoxia or anemia. In patients with chronic renal failure, impaired kidney function leads to significant reductions in EPO production, the primary cause of renal anemia. Exogenous EPO supplementation effectively compensates for this hematopoietic factor deficiency, restoring normal erythropoiesis.
4.1.3 Pharmacokinetic Characteristics
Following intravenous injection of EPO (150 International Units/kg), blood concentrations peak rapidly at 3,000–5,000 International Units. In long-term hemodialysis patients, the half-life after a single dose is approximately 8–10 hours, shortening to around 6 hours with repeated administration. Subcutaneous injection results in a delayed peak concentration (5–8 hours post-administration) but sustains effective levels for a longer duration, though its bioavailability is only about 20% of that with intravenous injection. EPO is primarily metabolized in the liver, with merely 10% excreted unchanged by the kidneys. Clinically, hematocrit increases positively correlate with dosage during the initial 1–3 weeks of treatment, typically reaching the target range of 30%–40% within 4–6 weeks, significantly enhancing patients' exercise tolerance, emotional well-being, and quality of life.
4.2 Research Methods Related to EPO
Research on EPO encompasses a range of in vitro and in vivo approaches, supported by laboratory testing and clinical evaluation:
In Vitro Experiments: Include cell culture studies to assess EPO's effects on the proliferation and differentiation of erythroid progenitor cells (e.g., CFU-E, BFU-E). Techniques such as flow cytometry, Western blotting, and quantitative real-time PCR are used to analyze cellular responses, signaling pathway activation, and gene expression changes.
In Vivo Models: Animal models (e.g., mouse models of renal anemia, hypoxia-induced anemia) are employed to investigate EPO's physiological and therapeutic effects in living organisms. These models help evaluate EPO's efficacy in improving hematological parameters, tissue oxygenation, and overall survival.
Pharmacokinetic Studies: Involve measuring EPO concentrations in blood or tissue samples at various time points following administration to determine absorption, distribution, metabolism, and excretion profiles. Techniques such as enzyme-linked immunosorbent assay (ELISA) and high-performance liquid chromatography (HPLC) are commonly used for quantitative analysis.
Clinical Trials: Evaluate EPO's safety and efficacy in human subjects, including studies on dosage optimization, treatment response, and long-term outcomes. Clinical parameters such as hemoglobin levels, hematocrit, blood pressure, and adverse events are closely monitored.
Quality Control Assays: For EPO-based biological products, quality control methods include purity analysis (e.g., SDS-PAGE, SEC-HPLC), endotoxin testing, and biological activity assays (e.g., cell proliferation assays) to ensure product consistency and reliability.
4.3 Application of ANT BIO PTE. LTD. Products in Research
ANT BIO PTE. LTD., through its sub-brand UA, delivers high-quality recombinant EPO proteins that are indispensable for advancing EPO-related research and drug development. The "EPO Protein, Human" (Cat No: UA040004) is a flagship product, characterized by exceptional biological activity, high purity, and excellent stability. Produced using advanced recombinant expression technology (CHO expression system), this product offers reliable performance across diverse research scenarios.
Key applications of this product include:
Cell Proliferation and Differentiation Studies: It is widely used for in vitro induction of hematopoietic stem cell differentiation into the erythroid lineage, enabling researchers to dissect the molecular mechanisms underlying erythropoiesis and evaluate the effects of various factors on this process.
Drug Development and Screening: Serves as a critical component in cell culture systems for the efficacy evaluation and high-throughput screening of erythropoiesis-stimulating drugs. Its consistent biological activity ensures accurate and reproducible results, accelerating the drug discovery process.
Quality Control of Biological Products: Functions as a reference standard for determining the activity and content of EPO-based biological drugs, ensuring compliance with industry quality standards and regulatory requirements.
Anemia Disease Model Research: Facilitates the construction of in vitro and in vivo anemia models, providing a valuable platform for testing the therapeutic potential of novel treatments targeting EPO signaling or erythropoiesis.
Additionally, ANT BIO PTE. LTD. offers the "Mouse EPO Protein, His tag" (Cat No: S0A4077) through its Starter sub-brand, which is ideal for mouse-related hematopoietic mechanism research, disease model construction, and target interaction studies.
5. Brand Mission
ANT BIO PTE. LTD. is dedicated to empowering the global life science community by providing high-quality, high-value biological reagents and innovative solutions. With a strong commitment to excellence, the company leverages advanced technology platforms—including recombinant antibody development (recombinant rabbit monoclonal, recombinant mouse monoclonal, rapid mouse monoclonal), recombinant protein expression systems (E.coli, CHO, HEK293, Insect Cells), One-Step ELISA Platform, and PTM Pan-Modification Antibody Platform—to develop a comprehensive portfolio of products, including antibodies, proteins, ELISA kits, and general life science reagents.
Through its three specialized sub-brands—Absin (focusing on general reagents and kits), Starter (specializing in antibodies), and UA (concentrating on recombinant proteins)—ANT BIO PTE. LTD. caters to the diverse needs of researchers in academia and biopharmaceutical industry. The company’s mission is to accelerate scientific discovery and advance global healthcare by delivering reliable, cutting-edge research tools and exceptional technical support. Having successfully obtained EU 98/79/EC certification, ISO9001 certification, and ISO13485 certification, ANT BIO PTE. LTD. adheres to the highest quality standards, ensuring that every product meets the rigorous demands of life science research and drug development.
6. Related Product List
| Product Code |
Product Name |
| UA040004 |
EPO Protein, Human |
| S0A4077 |
Mouse EPO Protein, His tag |
| S0A4076 |
Mycobacterium tuberculosis fbpB/Ag85B Protein, His Tag |
7. AI Disclaimer
This article is AI-compiled and interpreted based on the original work. All intellectual property (e.g., images, data) of the original publication shall belong to the journal and the research team. For any infringement, please contact us promptly and we will take immediate action.
ANT BIO PTE. LTD. – Empowering Scientific Breakthroughs
At ANTBIO, we are committed to advancing life science research through high-quality, reliable reagents and comprehensive solutions. Our specialized sub-brands (Absin, Starter, UA) cover a full spectrum of research needs, from general reagents and kits to antibodies and recombinant proteins. With a focus on innovation, quality, and customer-centricity, we strive to be your trusted partner in unlocking scientific mysteries and driving medical progress. Explore our product portfolio today and elevate your research to new heights.
|