Home > News > PCSK9 Antibodies: Pioneering Technological Revolutions and Clinical Breakthroughs in Lipid Management Therapy
PCSK9 Antibodies: Pioneering Technological Revolutions and Clinical Breakthroughs in Lipid Management Therapy
- 1. Literature Information
Research Focus: Exploration of PCSK9 (proprotein convertase subtilisin/kexin type 9) as a key target for lipid management, clinical applications of PCSK9-targeted therapies (including antibodies), technological innovations across drug platforms, and expanded therapeutic potential beyond lipid lowering.
Core Innovation: Establishing PCSK9 as a transformative target for cardiovascular therapy, with monoclonal antibodies leading the way and novel technologies (siRNA, gene editing) addressing clinical limitations—paving the path for personalized and long-term lipid management.
2. Research Background
Since statins emerged in the 1970s, they have remained the cornerstone of lipid-lowering therapy. However, their efficacy is limited in some patients, and unmet needs persist for more potent, convenient lipid management strategies. The discovery of PCSK9 marked a milestone: this protein binds to low-density lipoprotein receptors (LDL-R) on hepatocytes, promoting their lysosomal degradation and thereby increasing plasma LDL-C (low-density lipoprotein cholesterol) levels. Genome-wide association studies confirmed that loss-of-function PCSK9 mutations correlate with lower LDL-C and reduced cardiovascular disease risk, validating PCSK9 as a critical therapeutic target. PCSK9 antibodies (e.g., Repatha, Praluent) have since demonstrated remarkable clinical efficacy, but challenges such as injection administration and high costs have hindered widespread adoption. This research explores the technological evolution and clinical expansion of PCSK9-targeted therapies, highlighting the role of high-quality antibodies in advancing drug development and basic research.
3. Research Approaches
To advance PCSK9-targeted therapy, the research team adopted a translational, multi-platform strategy:
Mechanistic Validation: Investigating PCSK9’s interaction with LDL-R and its impact on cholesterol metabolism using biochemical and cellular assays.
Clinical Efficacy Assessment: Analyzing Phase III trial data of PCSK9 antibodies to evaluate LDL-C reduction, cardiovascular event prevention, and synergy with statins.
Technological Innovation Exploration: Evaluating novel platforms (siRNA, antisense oligonucleotides, gene editing) to address antibody limitations (administration, cost).
Indication Expansion Studies: Exploring PCSK9 inhibitors’ roles in cancer immunotherapy, metabolic syndrome, and other non-lipid-related diseases.
Comparative Platform Analysis: Assessing the advantages, limitations, and complementary roles of antibodies, RNAi, gene editing, and small molecules.
Pharmacodynamic Biomarker Development: Using PCSK9-specific antibodies to monitor target engagement and treatment response in preclinical and clinical studies.
4. Research Outcomes
4.1 PCSK9: A Transformative Target for Lipid Management
PCSK9 regulates LDL-C levels by mediating LDL-R degradation. Inhibition of PCSK9 (via antibodies or other modalities) increases LDL-R recycling to the hepatocyte surface, enhancing LDL-C clearance from the bloodstream. Loss-of-function PCSK9 mutations confirm this pathway’s physiological relevance, providing a strong scientific basis for targeted therapy.
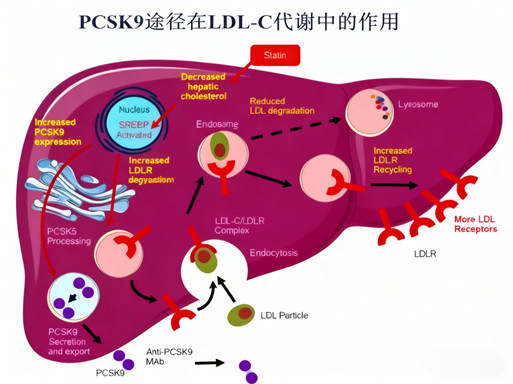
4.2 Clinical Performance and Challenges of PCSK9 Antibodies
Efficacy: PCSK9 antibodies reduce LDL-C by 50%-60% as monotherapy or in combination with statins, significantly lowering major adverse cardiovascular events in Phase III trials.
Limitations: Injection administration (burdensome for chronic disease patients) and high treatment costs have restricted market adoption, with 2020 global sales of leading products below $1 billion.
4.3 Technological Breakthroughs in Novel PCSK9-Targeted Drugs
To overcome antibody limitations, innovative platforms have emerged:
siRNA Technology: Inclisiran targets PCSK9 mRNA for long-term gene silencing, requiring only biannual subcutaneous injections; Phase III trials confirm sustained LDL-C reduction (>50%).
Antisense Oligonucleotides: AZD8233 (oral formulation) is in Phase II trials, offering potential for oral administration.
Gene Editing: CRISPR-Cas9 systems (delivered via lipid nanoparticles to the liver) enable permanent PCSK9 knockout; preclinical studies show single-dose, long-term lipid-lowering effects.
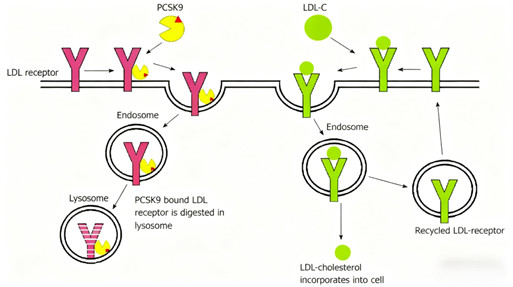
4.4 Expanded Clinical Applications Beyond Lipid Management
Cancer Immunotherapy: PCSK9 antibodies modulate immune cell function in the tumor microenvironment, enhancing PD-1 inhibitor efficacy and overcoming immunotherapy resistance in animal studies.
Metabolic Syndrome: Emerging roles in glucose metabolism and inflammation suggest potential for treating metabolic disorders beyond dyslipidemia.
Personalized Medicine: PCSK9 polymorphism-based stratification enables tailored lipid-lowering regimens for high-risk patients.
4.5 Comparative Advantages of Technology Platforms
| Platform | Key Advantages | Limitations |
| Antibodies | Immediate reversibility, established safety profile | Injection administration, high cost |
| RNAi | Reduced dosing frequency (biannual), improved compliance | Long-term safety data pending |
| Gene Editing | Potential "one-time, lifelong" treatment | Ethical concerns, long-term safety evaluation needed |
| Small Molecules | Oral administration potential, lower production costs | Early-stage development, limited efficacy data |
4.6 Future Directions for PCSK9-Targeted Therapies
Combination Strategies: Optimizing PCSK9 inhibitors with statins, ezetimibe, or other lipid-lowering agents.
Delivery Systems: Developing liver-targeted carriers to enhance efficacy and reduce off-target effects.
Indication Expansion: Exploring applications in Alzheimer’s disease, non-alcoholic fatty liver disease (NAFLD), and other lipid-related conditions.
Health Economics: Innovating production processes to reduce costs and improve accessibility.
5. Product Empowerment by ANT BIO PTE. LTD.
ANT BIO PTE. LTD.’s STARTER brand (antibodies) and UA brand (recombinant proteins) provide comprehensive tools for PCSK9 research and drug development. Key products include:
5.1 PCSK9 Antibodies (STARTER Brand)
* "PCSK9 Recombinant Rabbit mAb" (Catalog Nos.: S0B3245, S0B3246): High-specificity, sensitive antibodies validated for immunohistochemistry (IHC) and Western Blot (WB).
Roles in Research: Enabling PCSK9 expression profiling in liver tissues, mechanism studies of LDL-R degradation, and pharmacodynamic biomarker analysis for PCSK9 inhibitors.
Core Advantages: Precise cytoplasmic staining in FFPE samples, minimal background, and batch consistency—ensuring reliable results for preclinical and clinical studies.
5.2 PCSK9 Recombinant Proteins (UA Brand)
* Human (UA010997), Rat (UA010294), and Mouse (UA010261) PCSK9 His Tag Proteins (expressed in HEK293):
Roles in Research: Supporting antibody validation, binding assays (PCSK9-LDL-R interaction), and in vitro functional studies of PCSK9 inhibition.
These products are indispensable for basic research (lipid metabolism, cardiovascular disease mechanisms) and translational applications (drug discovery, pharmacodynamic evaluation), providing researchers with standardized, high-quality tools to accelerate PCSK9-targeted therapy development.
6. Brand Mission
ANT BIO PTE. LTD. is dedicated to empowering global life science advancement through three specialized sub-brands: ABSIN (general reagents, ELISA kits), STARTER (antibodies), and UA (recombinant proteins). Leveraging advanced development platforms—including recombinant rabbit/mouse monoclonal antibody generation, rapid antibody development, recombinant protein expression (E.coli, CHO, HEK293, Insect Cells), One-Step ELISA, and PTM Pan-Modification Antibody platforms—we deliver high-quality, compliant products certified by EU 98/79/EC, ISO9001, and ISO13485. Our mission is to partner with innovative pharmaceutical companies, research institutions, and scientists worldwide, providing innovative reagents and solutions that accelerate discoveries in cardiovascular medicine, metabolic diseases, and precision therapy.
7. Related Product List
| UA010997 | PCSK9 His Tag Protein, Human | Host : Human Expression System : HEK293 |
| UA010294 | PCSK9 His Tag Protein, Rat | Host : Rat Expression System : HEK293 Conjugation : Unconjugated |
| UA010261 | PCSK9 His Tag Protein, Mouse | Host : Mouse Expression System : HEK293 Conjugation : Unconjugated |
| S0B3245 | PCSK9 Recombinant Rabbit mAb (SDT-424-252) | Host : Rabbit Conjugation : Unconjugated |
| S0B3246 | PCSK9 Recombinant Rabbit mAb (SDT-424-16) | Host : Rabbit Conjugation : Unconjugated |
8. AI Disclaimer
This article is AI-compiled and interpreted based on the original work. All intellectual property (e.g., images, data) of the original publication shall belong to the journal and the research team. For any infringement, please contact us promptly and we will take immediate action.
ANT BIO PTE. LTD. – Empowering Scientific Breakthroughs
At ANTBIO, we are committed to advancing life science research through high-quality, reliable reagents and comprehensive solutions. Our specialized sub-brands (Absin, Starter, UA) cover a full spectrum of research needs, from general reagents and kits to antibodies and recombinant proteins. With a focus on innovation, quality, and customer-centricity, we strive to be your trusted partner in unlocking scientific mysteries and driving medical progress. Explore our product portfolio today and elevate your research to new heights.
Related News
- U.S. FDA Grants Breakthrough Device Designation to Laguna Diagnostics’mRNA Gene 4/30/2026
- Soluble fms-like Tyrosine Kinase-1 (sFlt-1): Pathophysiological Roles and Clinic 4/29/2026
- Mouse SHP Substrate 1: A Key Immune Regulator in Disease Models and Signal Trans 4/28/2026
- CMS Signed An Exclusive Commercialization and Supply Agreement with Pharmacosmos 4/28/2026
- Continuous Flow Porphyrin Synthesis Reveals New Insights Through Real-Time UV-Vi 4/27/2026
- SHPS1: A Pivotal Regulator of Immune Signaling, Tumor Evasion, and Beyond 4/27/2026
- The SHPS1 Mouse Model: Unraveling Immune Regulation, Tumor Evasion, and Neurolog 4/26/2026
- SHPS1 Protein: A Multifunctional Cell Surface Receptor Shaping Immunity, Develop 4/25/2026
- CMS: NDA for the Seasonal Allergic Rhinitis Indication of Class 1 Innovative Dru 4/24/2026
- TBK1 Antibodies: A Promising Therapeutic Avenue for Neurodegenerative Diseases 4/23/2026


