Home > News > Glycosylation Modification of PYCR1: Reshaping the Biosynthetic Pathways of Natural Products
Glycosylation Modification of PYCR1: Reshaping the Biosynthetic Pathways of Natural Products
- 1. Concept
Elucidating the biosynthetic mechanisms of highly active natural products with consecutive stereocenters and polycyclic systems has long been hindered by technical bottlenecks. These complex molecules typically feature carbon skeletons constructed via [4+2] cycloaddition reactions, but the enzymatic mechanisms driving these key steps remain poorly understood. PYCR1 (pyrroline-5-carboxylate reductase 1), a biosynthetic enzyme, undergoes glycosylation—a widespread eukaryotic post-translational modification. This modification redefines PYCR1’s catalytic capabilities, enabling efficient tandem [4+2] cycloaddition reactions that are critical for completing the total synthesis of complex natural products. As such, PYCR1’s glycosylation provides a paradigm for understanding how post-translational modifications regulate enzyme function and reshape natural product biosynthesis.
2. Research Frontiers
2.1 Fundamental Challenges in Complex Natural Product Biosynthesis
Biosynthesis of complex natural products faces unique obstacles:
* Limited Catalytic Efficiency of Non-Glycosylated Enzymes: Prokaryotically expressed non-glycosylated enzymes can only mediate single cyclization reactions with low efficiency, failing to drive the full synthesis of complex molecules.
* Poorly Understood Enzymatic Mechanisms: The enzymes catalyzing key [4+2] cycloaddition reactions in complex carbon skeleton formation have remained elusive.
* Knowledge Gaps in Post-Translational Regulation: How modifications like glycosylation influence biosynthetic enzyme function has not been fully explored, limiting the optimization of biosynthesis pathways.
2.2 Glycosylation Redefining the Functional Boundaries of Biosynthetic Enzymes
Glycosylation transforms PYCR1’s catalytic potential:
* Catalytic Efficiency Enhancement: Glycosylated PYCR1 exhibits orders of magnitude higher catalytic efficiency compared to its non-glycosylated counterpart.
* Novel Catalytic Capabilities: Glycosylation endows PYCR1 with the ability to perform tandem [4+2] cycloaddition reactions—a qualitative breakthrough not seen in non-glycosylated enzymes.
* Metabolic Evolution Insight: This functional expansion challenges traditional views of glycosylation (primarily associated with protein folding/stability) and highlights its role in expanding enzymatic catalytic scope during metabolic evolution.
2.3 Structural Basis and Innovative Catalytic Mechanisms of PYCR1
PYCR1, the first identified tandem [4+2] cyclase, features groundbreaking catalytic traits:
* Calcium-Dependent Catalysis: A unique mechanism where calcium ions play dual roles—structural support for the active site and direct promotion of catalytic reactions.
* Structural Elucidation: High-resolution crystal structures combined with quantum chemical calculations clarify the precise configuration of enzyme-substrate complexes and the structural basis of reaction selectivity.
* Key Residue Validation: Site-directed mutagenesis confirms the critical roles of specific amino acid residues in substrate recognition and catalysis, establishing a comprehensive catalytic model.
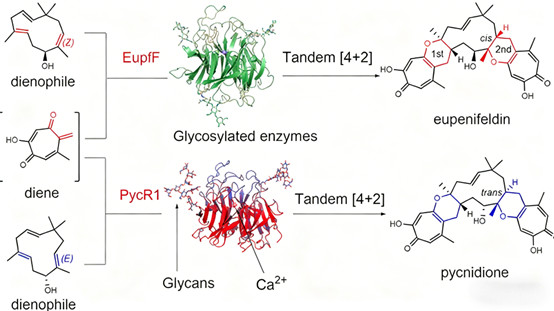
2.4 Synergistic Mechanism Between Glycosylation and Metal Cofactors
The synergistic interaction between glycosylation and calcium ions is pivotal to PYCR1’s function:
* Enhanced Cofactor Binding: Glycans increase the enzyme’s affinity for calcium ions, stabilizing the geometric configuration of the catalytic center.
* Conformational Rearrangement: Glycosylation induces structural changes in PYCR1, creating a microenvironment optimized for tandem cyclization reactions.
* Substrate Specificity Improvement: Indirect interactions between glycans and substrates enhance reaction specificity and efficiency, enabling consecutive catalytic steps.
2.5 Technological and Platform Breakthroughs
This research achieves multiple technical milestones:
* Eukaryotic Heterologous Expression System: Enables production of functionally intact PYCR1 with complete post-translational glycosylation.
* Multidisciplinary Research Strategy: Integrates bioinformatics, protein mass spectrometry, in vitro biochemical validation, and computational simulation to dissect enzyme function.
* End-to-End Technological Chain: Covers enzyme function discovery, structural characterization, and large-scale natural product synthesis, laying the groundwork for pharmacological studies.
2.6 Impact on Natural Product Research
This discovery transforms natural product research across multiple dimensions:
* Fundamental Science: Redefines understanding of glycosylation’s functional roles, opening new avenues for studying post-translational modification in enzyme regulation.
* Methodology: Establishes novel biosynthesis strategies based on glycosylated enzymes, enabling efficient production of complex natural products.
* Application: Provides new platforms and design principles for natural drug discovery and optimization, driving innovation in drug development.
3. Research Significance
PYCR1’s glycosylation-mediated functional expansion addresses a critical bottleneck in complex natural product biosynthesis. By revealing how glycosylation enhances catalytic efficiency and enables novel tandem reactions, this research provides a new theoretical framework for understanding enzyme regulation via post-translational modifications. The technological breakthroughs (e.g., eukaryotic expression, multidisciplinary characterization) enable large-scale synthesis of complex natural products, accelerating drug discovery and development. Furthermore, this paradigm encourages systematic exploration of glycosylation’s role in other biosynthetic enzymes, potentially unlocking new biosynthetic pathways for diverse natural products and advancing synthetic biology.
4. Related Mechanisms, Research Methods, and Product Applications
4.1 Mechanisms
PYCR1’s glycosylation drives natural product biosynthesis through two core mechanisms:
* Synergistic Catalysis: Glycosylation and calcium ions work together to stabilize the catalytic center, enhance substrate binding, and promote consecutive [4+2] cycloaddition reactions.
* Functional Expansion: Glycosylation-induced conformational changes expand PYCR1’s catalytic scope, enabling it to mediate multi-step reactions required for complex carbon skeleton formation.
4.2 Research Methods
Key methods for studying PYCR1 and glycosylation include:
* Heterologous Expression: Eukaryotic expression systems (e.g., CHO, HEK293) to produce glycosylated PYCR1; prokaryotic expression for non-glycosylated controls.
* Structural Biology: X-ray crystallography to determine high-resolution enzyme structures and enzyme-substrate complexes.
* Biochemical Assays: In vitro catalytic activity assays to measure efficiency and product formation; mass spectrometry to confirm glycosylation modification.
* Computational Simulation: Quantum chemical calculations and molecular dynamics simulations to model catalytic mechanisms and glycosylation effects.
4.3 Product Applications
ANT BIO PTE. LTD.’s "PYCR1 Rabbit Polyclonal Antibody" (Catalog No.: S0B1480) is a high-quality tool for studying PYCR1’s role in metabolism and biosynthesis:
* Cancer Metabolism Research: Investigates PYCR1’s function in proline metabolism, supporting cancer cell proliferation and oxidative stress resistance (e.g., liver cancer, prostate cancer, melanoma).
* Skin Biology Research: Explores PYCR1’s involvement in collagen synthesis, wrinkle formation, and genetic disorders like cutis laxa.
* Oxidative Stress Regulation: Studies PYCR1’s role in maintaining intracellular redox homeostasis.
* Metabolic Pathway Crosstalk: Analyzes interactions between proline metabolism and other pathways (e.g., glutamine metabolism).
The S0B1480 antibody offers exceptional advantages: high specificity with clear mitochondrial localization (ensuring accurate PYCR1 detection) and outstanding affinity/batch consistency—critical for reliable results in long-term research projects.
5. Brand Mission
ANT BIO PTE. LTD. is dedicated to empowering the global life science community with high-quality, innovative biological reagents and solutions. Leveraging advanced development platforms—including recombinant rabbit monoclonal antibody, recombinant mouse monoclonal antibody, rapid monoclonal antibody, and multi-system recombinant protein expression platforms (E.coli, CHO, HEK293, Insect Cells)—and adhering to rigorous international certifications (EU 98/79/EC, ISO9001, ISO13485), we strive to deliver reliable, performance-proven tools that accelerate scientific breakthroughs in synthetic biology, metabolism, and drug development. Our commitment to quality and innovation aims to support researchers and clinicians in advancing human health through cutting-edge life science research.
6. Related Product List
| Catalog No. | Product Name | Host |
| S0B1480 | PYCR1 Rabbit Polyclonal Antibody | Rabbit |
7. AI Disclaimer
This article is AI-compiled and interpreted based on the original work. All intellectual property (e.g., images, data) of the original publication shall belong to the journal and the research team. For any infringement, please contact us promptly and we will take immediate action.
ANT BIO PTE. LTD. – Empowering Scientific Breakthroughs
At ANTBIO, we are committed to advancing life science research through high-quality, reliable reagents and comprehensive solutions. Our specialized sub-brands (Absin, Starter, UA) cover a full spectrum of research needs, from general reagents and kits to antibodies and recombinant proteins. With a focus on innovation, quality, and customer-centricity, we strive to be your trusted partner in unlocking scientific mysteries and driving medical progress. Explore our product portfolio today and elevate your research to new heights.
Related News
- U.S. FDA Grants Breakthrough Device Designation to Laguna Diagnostics’mRNA Gene 4/30/2026
- Soluble fms-like Tyrosine Kinase-1 (sFlt-1): Pathophysiological Roles and Clinic 4/29/2026
- Mouse SHP Substrate 1: A Key Immune Regulator in Disease Models and Signal Trans 4/28/2026
- CMS Signed An Exclusive Commercialization and Supply Agreement with Pharmacosmos 4/28/2026
- Continuous Flow Porphyrin Synthesis Reveals New Insights Through Real-Time UV-Vi 4/27/2026
- SHPS1: A Pivotal Regulator of Immune Signaling, Tumor Evasion, and Beyond 4/27/2026
- The SHPS1 Mouse Model: Unraveling Immune Regulation, Tumor Evasion, and Neurolog 4/26/2026
- SHPS1 Protein: A Multifunctional Cell Surface Receptor Shaping Immunity, Develop 4/25/2026
- CMS: NDA for the Seasonal Allergic Rhinitis Indication of Class 1 Innovative Dru 4/24/2026
- TBK1 Antibodies: A Promising Therapeutic Avenue for Neurodegenerative Diseases 4/23/2026


