Home > News > Advancing Pathological Diagnosis of Epithelial-Derived Tumors: Precision Detection with CK-HMW Antibodies
Advancing Pathological Diagnosis of Epithelial-Derived Tumors: Precision Detection with CK-HMW Antibodies
- 1. Concept
Cytokeratins are key members of the intermediate filament protein family in eukaryotic cell cytoskeletons, encompassing 54 known members with molecular weights ranging from 40–68 kDa. They are classified into Type I (acidic) and Type II (neutral to alkaline) based on isoelectric points and molecular weights. Under physiological conditions, specific Type I and Type II cytokeratins form heterodimers that assemble into tissue-specific intermediate filament networks. High molecular weight cytokeratins (CK-HMW)—including CK1–6, CK9, and CK10—exhibit typical cytoplasmic localization in immunohistochemical detection. As specific markers for epithelial-derived tissues, CK-HMW antibodies enable precise identification and classification of epithelial-derived tumors, serving as a cornerstone for advancing pathological diagnostic accuracy.
2. Research Frontiers
2.1 Molecular Characteristics and Biological Functions of Cytokeratins
Cytokeratins’structural and functional properties underpin their diagnostic utility:
* Heterodimer Assembly: Strict pairing of Type I and Type II cytokeratins forms stable heterodimers, which polymerize into intermediate filaments providing mechanical support to epithelial cells.
* Tissue-Specific Expression: Different epithelial tissues express unique cytokeratin profiles—CK-HMW is predominantly expressed in stratified squamous epithelium and basal cells of glandular tissues, enabling tissue origin identification of tumors.
* Structural Integrity Maintenance: Cytokeratin networks preserve cell shape and integrity, particularly in tissues subjected to mechanical stress, while their dysregulation in tumors reflects epithelial dedifferentiation.
2.2 Core Value of CK-HMW in Prostate Pathological Diagnosis
CK-HMW is indispensable for distinguishing benign and malignant prostate lesions:
* Basal Cell Layer Evaluation: The integrity of the prostate basal cell layer is a key diagnostic criterion—persistent CK-HMW expression indicates an intact basal cell layer, excluding invasive carcinoma.
* Differential Diagnosis Enhancement: Combined detection with P63 (another basal cell marker) and P504S (a prostate cancer marker) significantly improves the accuracy of diagnosing prostatic hyperplasia, atypical lesions, and prostate cancer.
* Clinical Decision Support: Objective CK-HMW expression patterns provide reliable pathological evidence for clinical treatment planning, including active surveillance or radical therapy.
2.3 Role of CK-HMW in Differential Diagnosis of Urinary System Tumors
CK-HMW enables precise stratification of urothelial lesions:
* Dysplasia vs. Carcinoma in Situ Differentiation: In urothelial dysplasia, CK-HMW is restricted to basal layer cells; in carcinoma in situ, it shows diffuse strong positivity across the entire epithelial layer.
* Biological Behavior Reflection: This differential expression pattern mirrors fundamental differences in lesion aggressiveness, guiding clinical decisions such as surveillance frequency or surgical intervention.
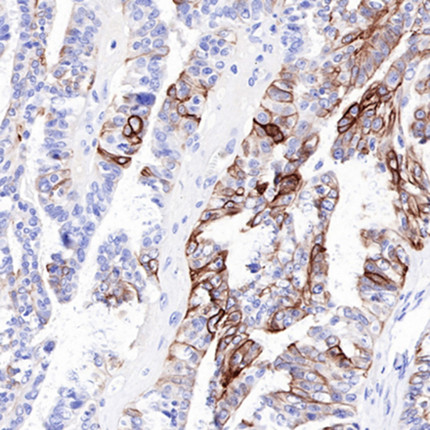
2.4 Multi-Marker Combined Detection Strategy for Optimizing Diagnostic Efficacy
Integrating CK-HMW with other markers enhances tumor diagnostic specificity and sensitivity:
* Thyroid Pathology: Combined detection with P53, Ki-67, and galectin-3 improves differentiation between papillary thyroid carcinoma and benign nodules (e.g., follicular adenoma).
* Epithelial Tumor Classification: Synergistic use of CK-HMW and low molecular weight cytokeratins (CK-LMW) reliably distinguishes squamous epithelial tumors (CK-HMW-positive) from non-squamous epithelial tumors (CK-LMW-positive).
* Broad Applicability: This combined approach has proven valuable across various epithelial-derived tumors, reducing diagnostic ambiguity and inter-observer variability.
2.5 Standardized Quality Control Systems Ensuring Test Result Reliability
Reliable CK-HMW detection depends on rigorous quality control:
* Limitations of Traditional Controls: Tissue-based controls suffer from long preparation cycles and high heterogeneity, leading to inconsistent quality assessment.
* Technological Breakthrough with Liquid Cell-Based Controls: Engineered cell line-derived liquid quality control products offer uniform, stable biomarker expression, enabling standardized internal laboratory quality control and inter-laboratory proficiency testing.
* Consistency Guarantee: Standardized quality control systems ensure comparability and reproducibility of CK-HMW results across different laboratories and test batches.
2.6 Technological Innovation Driving Precision in Pathological Diagnosis
Advancements in diagnostic technology elevate CK-HMW’s clinical utility:
* Liquid Cell-Based Technology: Addresses challenges such as rare positive sample scarcity and tissue heterogeneity, enabling standardized testing processes and objective result interpretation.
* AI Integration: Standardized quality control supports accurate digital pathology image analysis and remote diagnosis, reducing human error and improving diagnostic efficiency.
* Precision Development: Technological innovation drives pathological diagnosis toward higher standardization, objectivity, and personalized medicine alignment.
3. Research Significance
CK-HMW antibodies address a critical need in pathological diagnosis by enabling precise identification and classification of epithelial-derived tumors. Their role in evaluating prostate basal cell integrity, differentiating urinary system lesions, and supporting multi-marker combined detection significantly improves diagnostic accuracy and reduces misdiagnosis rates. Standardized quality control systems and technological innovations further enhance result reliability and comparability, laying the groundwork for consistent clinical decision-making. Beyond diagnosis, CK-HMW’s tissue-specific expression aids in tumor staging and prognosis assessment, supporting personalized treatment strategy formulation. As a core tool in modern pathology, CK-HMW advances the precision and efficiency of epithelial-derived tumor diagnosis, ultimately improving patient outcomes.
4. Related Mechanisms, Research Methods, and Product Applications
4.1 Mechanisms
CK-HMW’s diagnostic value stems from its epithelial-specific expression:
* Tissue Origin Marker: CK-HMW is exclusively expressed in epithelial cells and their derivatives, enabling distinction between epithelial and non-epithelial tumors (e.g., mesenchymal tumors).
* Differentiation Status Indicator: Reduced or absent CK-HMW expression in tumors reflects epithelial-mesenchymal transition (EMT), a marker of increased invasiveness and poor prognosis.
4.2 Research Methods
Key methods for CK-HMW research and application include:
* Immunohistochemistry (IHC): Primary detection method for CK-HMW in formalin-fixed paraffin-embedded (FFPE) samples, enabling qualitative and semi-quantitative analysis of expression patterns.
* Multi-Marker Panel Testing: Combined staining with P63, P504S, CK-LMW, and other markers to improve diagnostic specificity.
* Quality Control Validation: Use of liquid cell-based controls to verify staining performance, ensuring result reliability.
* Digital Pathology: AI-assisted image analysis for objective quantification of CK-HMW expression, reducing inter-observer variability.
4.3 Product Applications
ANT BIO PTE. LTD.’s CK-HMW antibodies, represented by the STARTER brand’s "S-RMab® High Molecular Weight Cytokeratin Recombinant Rabbit Monoclonal Antibody" (Catalog No.: S0B2119), are high-performance tools for pathological diagnosis:
* Squamous Cell Carcinoma Diagnosis: Specific identification of squamous cell carcinoma (e.g., lung, esophageal, cervical) and differentiation from adenocarcinoma or undifferentiated carcinoma.
* Myoepithelial Cell Marking: Labeling myoepithelial cells in breast, salivary gland, and other tissues to distinguish invasive carcinoma from carcinoma in situ.
* Prostate Basal Cell Marking: Assisting in differentiation between benign prostatic hyperplasia and prostate cancer by evaluating basal cell layer integrity.
* Thymoma Subtyping: Marking epithelial components in thymomas to support histological subtyping and prognosis assessment.
The S0B2119 antibody, developed using ANT BIO PTE. LTD.’s proprietary S-RMab® recombinant rabbit monoclonal platform and validated for IHC, offers exceptional advantages: high specificity for CK-HMW (e.g., CK5/6, CK14) with clear cytoplasmic localization (ensuring reliable epithelial cell identification in FFPE samples) and superior staining stability with minimal batch variation—critical for consistent results in clinical diagnostics and translational research.
5. Brand Mission
ANT BIO PTE. LTD. is dedicated to empowering the global life science community with high-quality, innovative biological reagents and solutions. Leveraging advanced development platforms—including recombinant rabbit monoclonal antibody, recombinant mouse monoclonal antibody, rapid monoclonal antibody, and multi-system recombinant protein expression platforms (E.coli, CHO, HEK293, Insect Cells)—and adhering to rigorous international certifications (EU 98/79/EC, ISO9001, ISO13485), we strive to deliver reliable, performance-proven tools that accelerate scientific breakthroughs in pathology, oncology, and translational medicine. Our commitment to quality and innovation aims to support researchers and clinicians in advancing human health through precise diagnosis and cutting-edge life science research.
6. Related Product List
| Catalog No. | Product Name | Host |
| S0B2119 | S-RMab® CK-HMW Recombinant Rabbit mAb (SDT-P002) | Rabbit |
| S0B2119P | S-RMab® CK-HMW Recombinant Rabbit mAb, PBS Only (SDT-P002) | Rabbit |
7. AI Disclaimer
This article is AI-compiled and interpreted based on the original work. All intellectual property (e.g., images, data) of the original publication shall belong to the journal and the research team. For any infringement, please contact us promptly and we will take immediate action.
ANT BIO PTE. LTD. – Empowering Scientific Breakthroughs
At ANTBIO, we are committed to advancing life science research through high-quality, reliable reagents and comprehensive solutions. Our specialized sub-brands (Absin, Starter, UA) cover a full spectrum of research needs, from general reagents and kits to antibodies and recombinant proteins. With a focus on innovation, quality, and customer-centricity, we strive to be your trusted partner in unlocking scientific mysteries and driving medical progress. Explore our product portfolio today and elevate your research to new heights.
Related News
- U.S. FDA Grants Breakthrough Device Designation to Laguna Diagnostics’mRNA Gene 4/30/2026
- Soluble fms-like Tyrosine Kinase-1 (sFlt-1): Pathophysiological Roles and Clinic 4/29/2026
- Mouse SHP Substrate 1: A Key Immune Regulator in Disease Models and Signal Trans 4/28/2026
- CMS Signed An Exclusive Commercialization and Supply Agreement with Pharmacosmos 4/28/2026
- Continuous Flow Porphyrin Synthesis Reveals New Insights Through Real-Time UV-Vi 4/27/2026
- SHPS1: A Pivotal Regulator of Immune Signaling, Tumor Evasion, and Beyond 4/27/2026
- The SHPS1 Mouse Model: Unraveling Immune Regulation, Tumor Evasion, and Neurolog 4/26/2026
- SHPS1 Protein: A Multifunctional Cell Surface Receptor Shaping Immunity, Develop 4/25/2026
- CMS: NDA for the Seasonal Allergic Rhinitis Indication of Class 1 Innovative Dru 4/24/2026
- TBK1 Antibodies: A Promising Therapeutic Avenue for Neurodegenerative Diseases 4/23/2026


